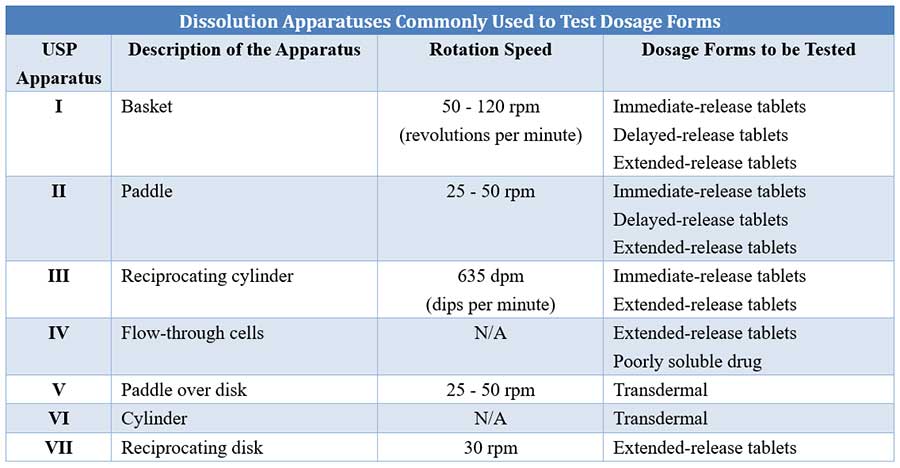
![PDF] DISSOLUTION PROFILE AND DRUG RELEASE KINETICS OF THREE SPECIALLY FORMULATED THEOPHYLLINE ENTERIC COATED SOLID DOSAGE FORM: A COMPARATIVE STUDY | Semantic Scholar PDF] DISSOLUTION PROFILE AND DRUG RELEASE KINETICS OF THREE SPECIALLY FORMULATED THEOPHYLLINE ENTERIC COATED SOLID DOSAGE FORM: A COMPARATIVE STUDY | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/da03b727452dd470dfba06da90af9745e003ae02/4-Table1-1.png)
PDF] DISSOLUTION PROFILE AND DRUG RELEASE KINETICS OF THREE SPECIALLY FORMULATED THEOPHYLLINE ENTERIC COATED SOLID DOSAGE FORM: A COMPARATIVE STUDY | Semantic Scholar

Regulating the pH of bicarbonate solutions without purging gases: Application to dissolution testing of enteric coated tablets, pellets and microparticles - ScienceDirect
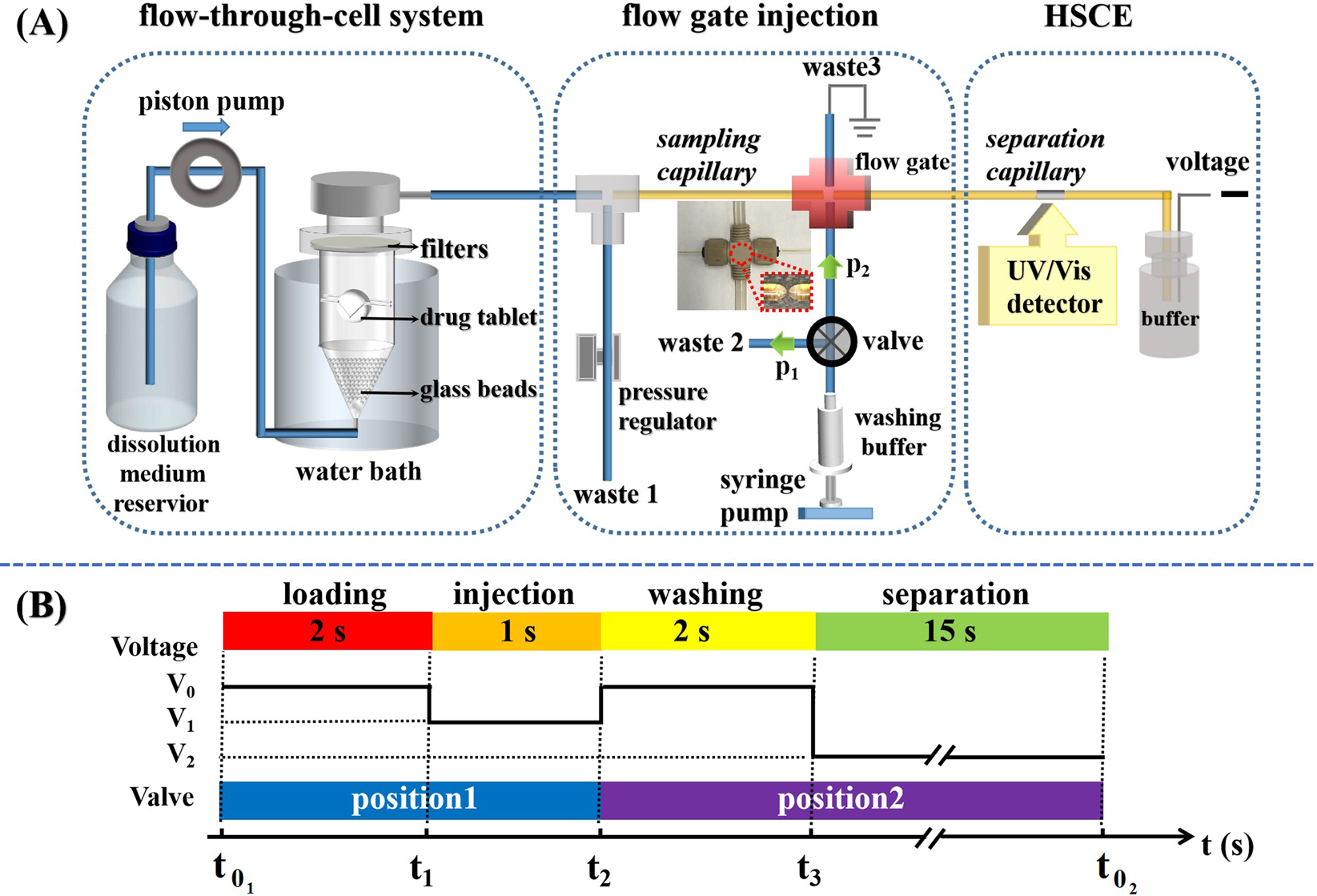
Automatic Dissolution Testing with High-Temporal Resolution for Both Immediate-Release and Fixed-Combination Drug Tablets | Scientific Reports
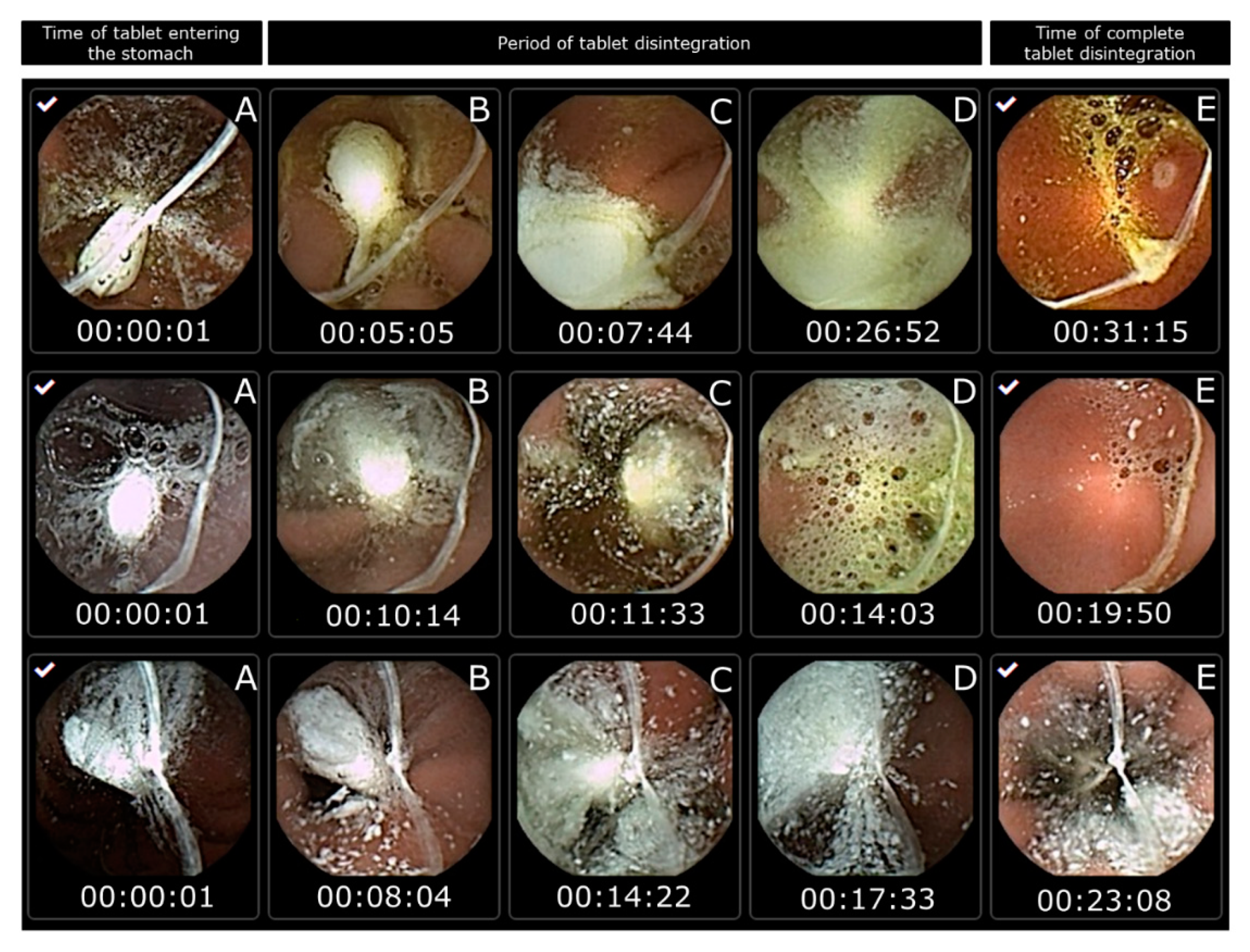
Pharmaceutics | Free Full-Text | The Use of Capsule Endoscopy to Determine Tablet Disintegration In Vivo

SOLVED: The In vitro Dissolution profile of levodopa controlled release tablets was done using 0.1 N HCl (900 mL) at 37 ± 0.5°C using USP dissolution apparatus II (Paddle) at 50 rounds
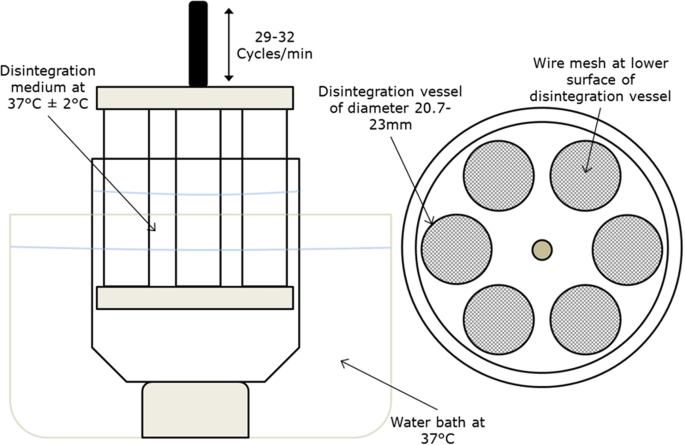
Conceptualisation, Development, Fabrication and In Vivo Validation of a Novel Disintegration Tester for Orally Disintegrating Tablets | Scientific Reports
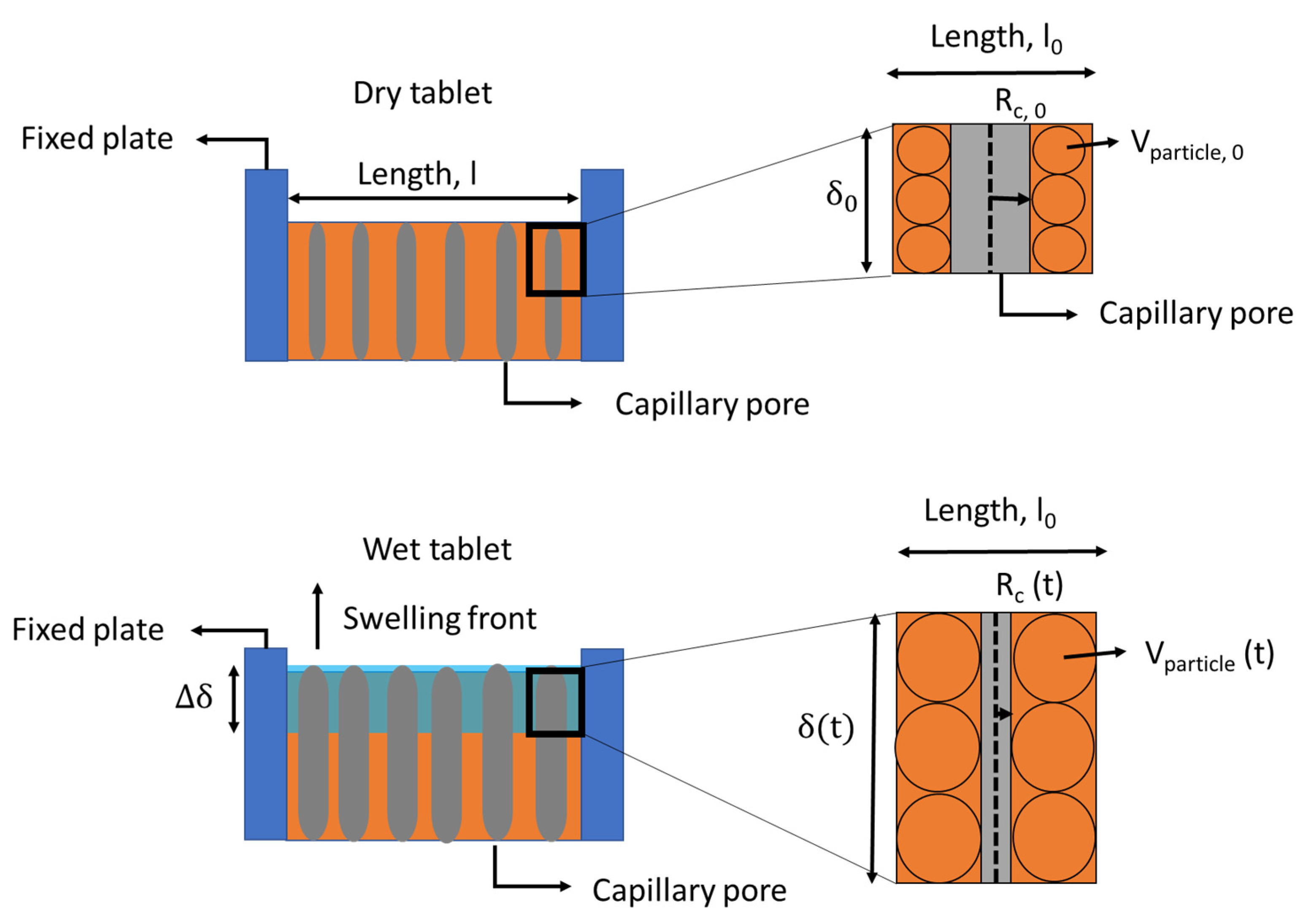
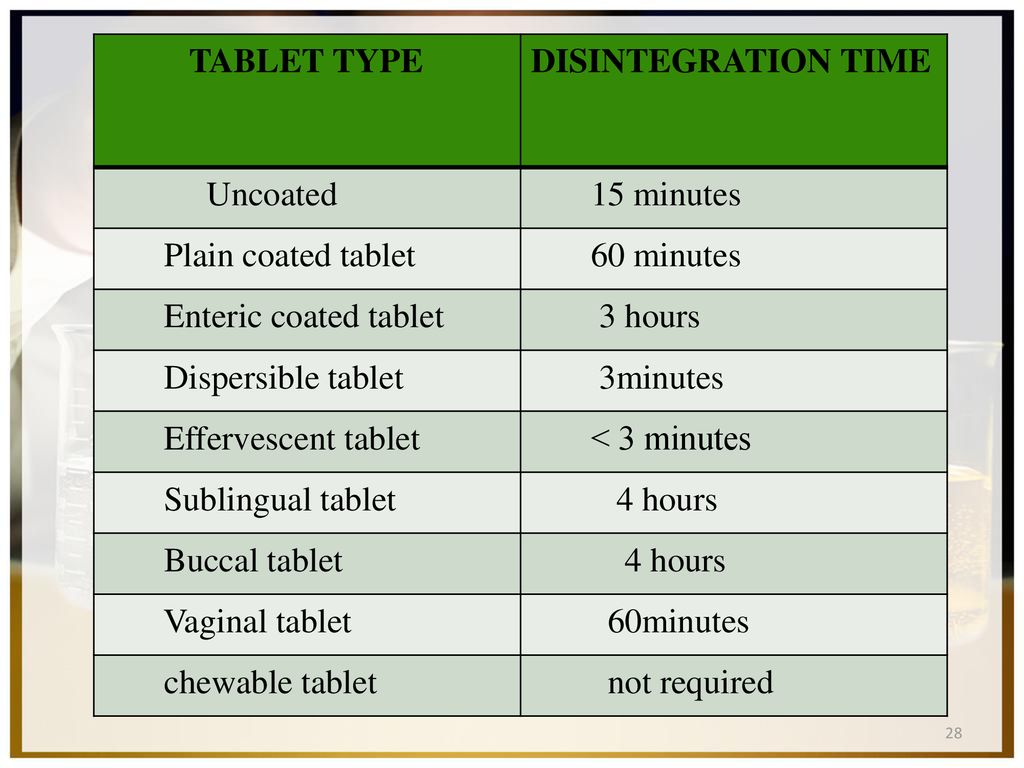

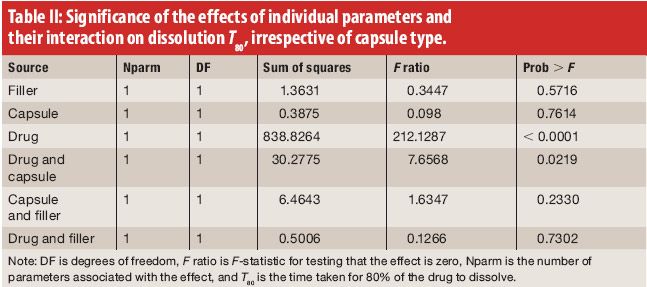


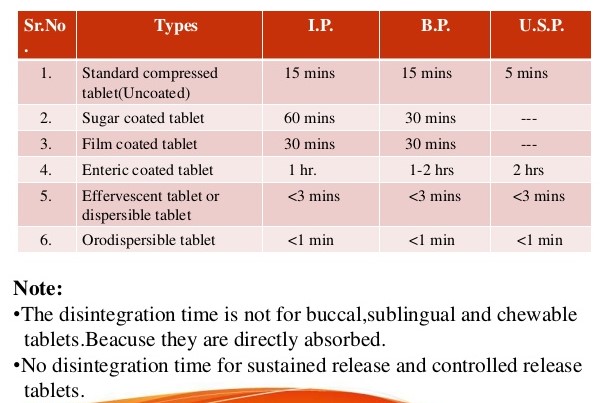
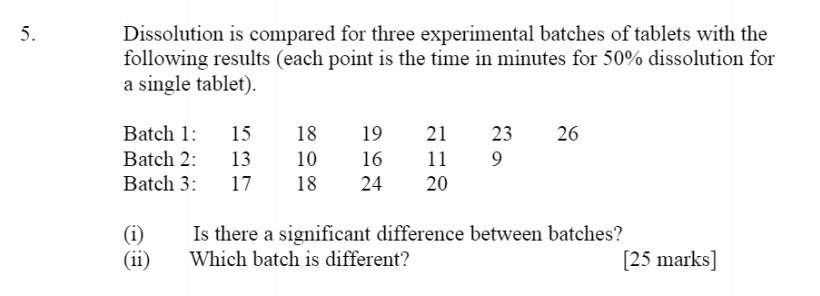
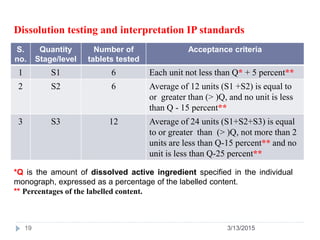


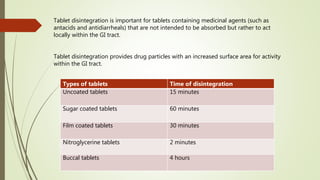
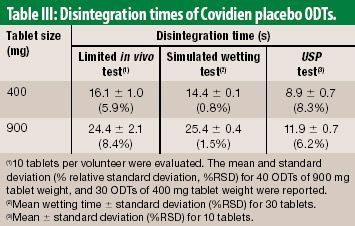
![PDF] The Significance of disintegration testing in pharmaceutical development | Semantic Scholar PDF] The Significance of disintegration testing in pharmaceutical development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/92a193f174837cd54c0ca8eb588ad0f3e936abc1/5-Table3-1.png)